SciMed Education
Mixing Calorimetry Methods and Applications
In Summary
Mixing calorimetry measures the heat released or absorbed when two or more substances come into contact under controlled temperature conditions. By monitoring the heat flow generated during the interaction, researchers can determine thermodynamic and kinetic information about reactions, adsorption processes and dissolution behaviour.
What is mixing calorimetry used for?
Mixing calorimetry is used to investigate the energetics of processes that occur when materials interact. This includes chemical reactions, adsorption of gases or liquids onto solids, dissolution processes and the mixing of liquids.
By measuring the heat released or absorbed during these interactions, scientists can determine enthalpy changes and gain insight into reaction mechanisms.
How are mixing calorimtery experiments performed?
In a typical mixing calorimetry experiment, the substances being studied are first stabilised at the same temperature inside the calorimeter.
They are then brought into contact within the calorimetric cell and the resulting heat flow is recorded from the moment the interaction begins.
The thermal signal provides information about the energy released or absorbed during the process.
How are mixing calorimtery experiments performed?
Several experimental configurations are used in mixing calorimetry. Batch mixing methods combine materials inside a sealed cell, often using mechanisms such as reversal or membrane mixing.
Flow mixing experiments introduce one reactant into another using controlled injection systems. Continuous flow or percolation methods allow gases or liquids to pass through solid samples while the resulting heat effects are measured.
What information can mixing calorimetry provide?
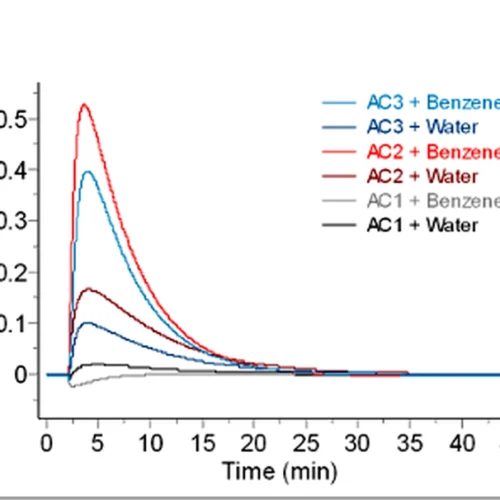
Mixing calorimetry directly measures the heat of interaction between substances. This can include heats of reaction, dissolution, adsorption or wetting.
Because the heat flow is recorded continuously, the technique can also provide insight into the kinetics of the process as well as the overall thermodynamic change.
What to do Next?
Mixing calorimetry provides valuable insight into the thermodynamic and kinetic behaviour of chemical interactions, including dissolution, adsorption and reaction processes. Instruments such as the MicroCalvet calorimeter enable researchers to measure these heat effects with exceptional sensitivity and precision.
If you would like to learn more about how mixing calorimetry can support your research or analytical applications, explore the MicroCalvet system below or contact the SciMed team for further guidance.
Page FAQ's
Yes. Specialised mixing cells can be designed to operate under elevated pressure conditions, allowing researchers to study gas liquid interactions or reactions involving pressurised systems.
Mixing calorimetry experiments generally use relatively small sample sizes so that thermal equilibrium can be reached quickly and accurate heat measurements can be obtained.
A wide range of materials can be analysed using mixing calorimetry including liquids, polymers, powders, catalysts and porous materials.
Contact Us Today
We take great pleasure in assisting you and ensuring you get a prompt response to your questions
Live chat opening hours Mon – Fri 9:15 to 16:30 (UK Time)
